2-Naphthacenecarboxamide, 4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl- 1,11-dioxo-, [4
S-(4

,4a

,5a

,6
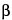
,12a

)]-.
(4
S,4a
S,5a
S,12a
S)-4-(Dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacene-carboxamide



[
60-54-8].
Trihydrate
498.49



[
6416-04-2].
Packaging and storage—
Preserve in tight, light-resistant containers.
Labeling—
Label it to indicate that it is to be used in the manufacture of nonparenteral drugs only.
Identification—
A:
Ultraviolet Absorption  197U
197U
—
Solution:
20 µg per mL.
Medium:
0.25 N sodium hydroxide.
Absorptivity 6 minutes after preparation, calculated on the anhydrous basis, at 380 nm is between 104.5% and 111.95% of that of USP Tetracycline Hydrochloric RS, the potency of the Reference Standard being taken into account.
B:
The chromatogram of the Assay obtained as directed in the Assay exhibits a major peak for tetracycline, the retention time of which corresponds to that exhibited in the chromatogram of the Standard preparation obtained as directed in the Assay.
C:
To 0.5 mg add 2 mL of sulfuric acid: a purplish red color is produced. Add the solution to 1 mL of water: the color becomes yellow.
D:
Prepare a
Test Solution in methanol containing the equivalent of 1 mg of tetracycline hydrochloride per mL, and proceed as directed for
Method II under
Identification—Tetracyclines  193
193
.
Specific rotation  781S
781S :
:
between

260

and

280

, calculated on the anhydrous basis.
Test solution:
5 mg per mL, in 0.1 N hydrochloric acid.
pH  791
791 :
:
between 3.0 and 7.0, in an aqueous suspension containing 10 mg per mL.
Heavy metals, Method II  231
231 :
:
0.005%.
Limit of 4-epianhydrotetracycline—
Using the
Diluting solvent,
Chromatographic system, and
Procedure set forth in the
Assay, chromatograph a Standard solution prepared by dissolving an accurately weighed quantity of
USP 4-Epianhydrotetracycline Hydrochloride RS in
Diluting solvent to obtain a solution having a known concentration of about 10 µg per mL. Using the chromatogram so obtained and the chromatogram of the
Assay preparation obtained as directed in the
Assay, calculate the percentage of 4-epianhydrotetracycline in the Tetracycline taken by the formula:
10(CE / W)(rU / rS),
in which
CE is the concentration, in µg per mL, of
USP 4-Epianhydrotetracycline Hydrochloride RS in the Standard solution,
W is the weight, in mg, of Tetracycline taken to prepare the
Assay preparation, and
rU and
rS are the 4-epianhydrotetracycline peak responses obtained from the
Assay preparation and the Standard solution, respectively: not more than 2.0% is found.
Assay—
Diluting solvent, Mobile phase, Standard preparation, Resolution solution, and Chromatographic system—
Prepare as directed in the
Assay under
Tetracycline Hydrochloride.
Assay preparation—
Transfer about 45 mg of Tetracycline, accurately weighed, to a 100-mL volumetric flask, dissolve in Diluting solvent, dilute with the same solvent to volume, and mix.
Procedure—
Proceed as directed in the
Assay under
Tetracycline Hydrochloride. Calculate the quantity, in µg, of tetracycline hydrochloride (C
22H
24N
2O
8·HCl) equivalent in each mg of Tetracycline taken by the formula:
100(CP / W)(rU / rS),
in which
W is the weight, in mg, of Tetracycline taken to prepare the
Assay preparation, and the other terms are as defined therein.