Many pharmaceutical substances are optically active in the sense that they rotate an incident plane of polarized light so that the transmitted light emerges at a measurable angle to the plane of the incident light. This property is characteristic of some crystals and of many pharmaceutical liquids or solutions of solids. Where the property is possessed by a liquid or by a solute in solution, it is generally the result of the presence of one or more asymmetric centers, usually a carbon atom with four different substituents. The number of optical isomers is 2n, where n is the number of asymmetric centers. Polarimetry, the measurement of optical rotation, of a pharmaceutical article may be the only convenient means for distinguishing optically active isomers from each other and thus is an important criterion of identity and purity.
Substances that may show optical rotatory power are
chiral. Those that rotate light in a clockwise direction as viewed towards the light source are
dextrorotatory, or (+)
optical isomers. Those that rotate light in the opposite direction are called
levorotatory or (–) optical isomers. (The symbols
d- and
l-, formerly used to indicate dextro- and levorotatory isomers, are no longer sanctioned owing to confusion with
D- and
L-, which refer to configuration relative to
D-glyceraldehyde. The symbols
R and
S and

and
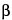
are also used to indicate configuration, the arrangement of atoms or groups of atoms in space.)
The physicochemical properties of nonsuperimposable chiral substances rotating plane polarized light in opposite directions to the same extent, enantiomers, are identical, except for this property and in their reactions with other chiral substances. Enantiomers often exhibit profound differences in pharmacology and toxicology, owing to the fact that biological receptors and enzymes themselves are chiral. Many articles from natural sources, such as amino acids, proteins, alkaloids, antibiotics, glycosides, and sugars, exist as chiral compounds. Synthesis of such compounds from nonchiral materials results in equal numbers of the enantiomers, racemates. Racemates have a net null optical rotation, and their physical properties may differ from those of the component enantiomers. Use of stereoselective or stereospecific synthetic methods or separation of racemic mixtures can be used to obtain individual optical isomers.
Measurement of optical rotation is performed using a polarimeter.
* The general equation used in polarimetry is:
where [

] is the specific rotation at wavelength
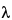
,
t is the temperature,
a is the observed rotation in degrees (

),
l is the pathlength in decimeters, and
c is the concentration of the analyte in g per 100 mL. Thus, [

] is 100 times the measured value, in degrees (

), for a solution containing 1 g in 100 mL, measured in a cell having a pathlength of 1.0 decimeter under defined conditions of incident wavelength of light and temperature. For some Pharmacopeial articles, especially liquids such as essential oils, the optical rotation requirement is expressed in terms of the observed rotation,
a, measured under conditions defined in the monograph.
Historically, polarimetry was performed using an instrument where the extent of optical rotation is estimated by visual matching of the intensity of split fields. For this reason, the D-line of the sodium lamp at the visible wavelength of 589 nm was most often employed. Specific rotation determined at the D-line is expressed by the symbol:
and much of the data available are expressed in this form. Use of lower wavelengths, such as those available with the mercury lamp lines isolated by means of filters of maximum transmittance at approximately 578, 546, 436, 405, and 365 nm in a photoelectric polarimeter, has been found to provide advantages in sensitivity with a consequent reduction in the concentration of the test compound. In general, the observed optical rotation at 436 nm is about double and at 365 nm about three times that at 589 nm. Reduction in the concentration of the solute required for measurement may sometimes be accomplished by conversion of the substance under test to one that has a significantly higher optical rotation. Optical rotation is also affected by the solvent used for the measurement, and this is always specified.
It is now common practice to use other light sources, such as xenon or tungsten halogen, with appropriate filters, because these may offer advantages of cost, long life, and broad wavelength emission range, over traditional light sources.
 Specific Rotation—
Specific Rotation—
The reference
Specific rotation  781S
781S
in a monograph signifies that specific rotation is to be calculated from observed optical rotations in the
Test solution obtained as directed therein. Unless otherwise directed, measurements of optical rotation are made at 589 nm at 25

. Where a photoelectric polarimeter is used, a single measurement, corrected for the solvent blank, is made. Where a visual polarimeter is employed, the average of no fewer than five determinations, corrected for the reading of the same tube with a solvent blank, is used. Temperature, which applies to the solution or the liquid under test, should be maintained within 0.5

of the stated value. Use the same cell for sample and blank. Maintain the same angular orientation of the cell in each reading. Place the cell so that the light passes through it in the same direction each time. Unless otherwise specified, specific rotation is calculated on the dried basis where
Loss on drying is specified in the monograph or on the anhydrous basis where
Water is specified.
Optical rotation of solutions should be determined within 30 minutes of preparation. In the case of substances known to undergo racemization or mutarotation, care should be taken to standardize the time between adding the solute to the Solvent and introduction of the solution into the polarimeter tube.
 Angular Rotation—
Angular Rotation—
The reference
Angular rotation  781A
781A
in a monograph signifies, unless otherwise directed, that the optical rotation of the neat liquid is measured in a 1.0-dm tube at 589 nm at 25

, corrected for the reading of the dry empty tube.
*
Suitable calibrators are available from the Office of Standard Reference Materials, National Institute of Standards and Technology (NIST), Gaithersburg, MD 20899, as current lots of Standard Reference Materials, Dextrose and Sucrose. Alternatively, calibration may be checked using a Polarization Reference Standard, which consists of a plate of quartz mounted in a holder perpendicular to the light path. These standards are available, traceable to NIST, from Rudolph Research Analytical, 354 Route 206, Flanders, NJ 07836, or from Rudolph Instruments, Inc., 40 Pier Lane, Fairfield, NJ 07004-2113.